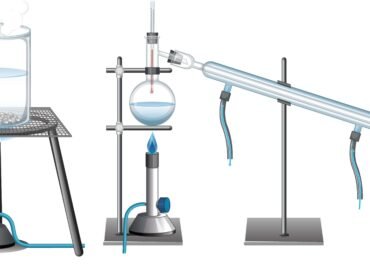
Have you ever wondered why scientists can separate a mixture of liquids that look the same? We stratify the two almost purely by fractional distillation.. The process requires you to obtain each fraction through your careful observation of temperature and your precise control of temperature.
Understanding The Identification of Fractions.
Understand every fraction at every distillation to get good separation. Each faction stands for a substance that boils at a specific temperature.
There are some important methods to distinguish various fractions:
Boiling Point Observation
- The boiling points of every liquid are different.
- The constant temperature measurement shows that you are measuring a particular fraction of the material.
- The temperature shift marks the beginning of a new fraction.
Thermometer Readings
- The thermometer is placed close to the top of the apparatus.
- A constant temperature means the collection of a pure fraction.
- The varying temperature is a sign of a mixture or passage between fractions.
Collection in Separate Containers.
-
Divide the mixtures into different conical flasks.
-
Upon mixing the solutions, it becomes mandatory to associate each flask with a specific temperature.
Physical Characteristics
- Fractions may vary in colour, odour, and viscosity.
- Such perceptible differences are helpful in identification.
Time of Collection
- Samples, at times are gathered at separate moments.
- Low-temperature fractions tend to boil at low temperatures.
Refractive Index or Density (Sophisticated Method)
- Refractive index or density is taken using lab instruments.
- These values prove the identity of all the fractions.
Chemical Tests
- Inspect a compound using specific chemical tests.
- This method helps separate materials with close boiling points.
Major points during Identification.
- Keep the heating rate constant to get the correct results.
- Note down the temperature and observations closely.
- Install the distillation equipment in a proper manner.
Conclusion
Detection of fractions in distillation combines observation and scientific measurement. We separate mixtures through three processes which include observing temperature changes and separating the liquid components and analysing their physical and chemical properties. It reflects the systematization of laboratories, which is much applied in petroleum refining, pharmaceuticals, chemicals, etc.
Need Customized Fractional Distillation Equipment?
Talk to our experts for tailored industrial solutions.
Contact us today: https://srilekhabioenvirotech.com/